Featured event
iScience PAPER PUBLISHED
JAN 3, 11:00 AM 2024
iScience PAPER PUBLISHED
JAN 3, 11:00 AM 2024
实验室软件著作获得授权
SEP 26, 11:00 AM 2023
Journal of Proteome Research Published
AUG 10, 11:00 AM 2023
实验室软件著作获得授权
MAY 31, 11:00 AM 2023
实验室软件著作获得授权
MAR 9, 11:00 AM 2023
PROGRESS IN NEUROBIOLOGY PAPER PUBLISHED
JAN 12, 11:00 AM 2023
祝贺牛孔艳获得中国科学院生物与化学交叉研究中心优秀研究生奖学金
NOV 25, 11:00 AM 2022
NAR PAPER PUBLISHED
NOV 21, 11:00 AM 2022
MCP PAPER PUBLISHED
AUG 26, 11:00 AM 2022
Nature Communications paper Published
June 20, 11:00 AM 2022
Cell Reports paper Published
June 2, 11:00 AM 2022
Genetics paper Published
Dec 21, 11:00 AM 2021
Molecular Psychiatry paper Published
OCT 25, 11:00 AM 2021
Cell Death and Disease paper Published
OCT 23, 11:00 AM 2021
Nature Communications paper Published
OCT 22nd, 11:00 AM 2021
实验室发明专利获得授权
SEP 26th, 11:00 AM 2021
Hepatology paper Published
June 5th, 11:00 AM 2021
Hepatology paper Published:Single-cell transcriptomic analysis reveals a hepatic stellate cell-activation roadmap and myofibroblast origin during liver fibrosis
iScience paper Published
April 10th, 11:00 AM 2021
iScience paper Published:Induced lineage promiscuity undermines the efficiency of all-trans-retinoid-acid-induced differentiation of acute myeloid leukemia
PNAS paper Published
March 1st, 11:00 AM 2021
PNAS paper Published:A RIPK1-regulated inflammatory microglial state in amyotrophic lateral sclerosis
PNAS paper Published
May 5th, 11:30 PM 2020
PNAS paper Published:Hepatocyte-Specific TAK1 Deficiency Drives RIPK1 Kinase-dependent Inflammation to Promote Liver Fibrosis and Hepatocellular Carcinoma
MCP paper Published
Aug 29th, 12:30 AM 2019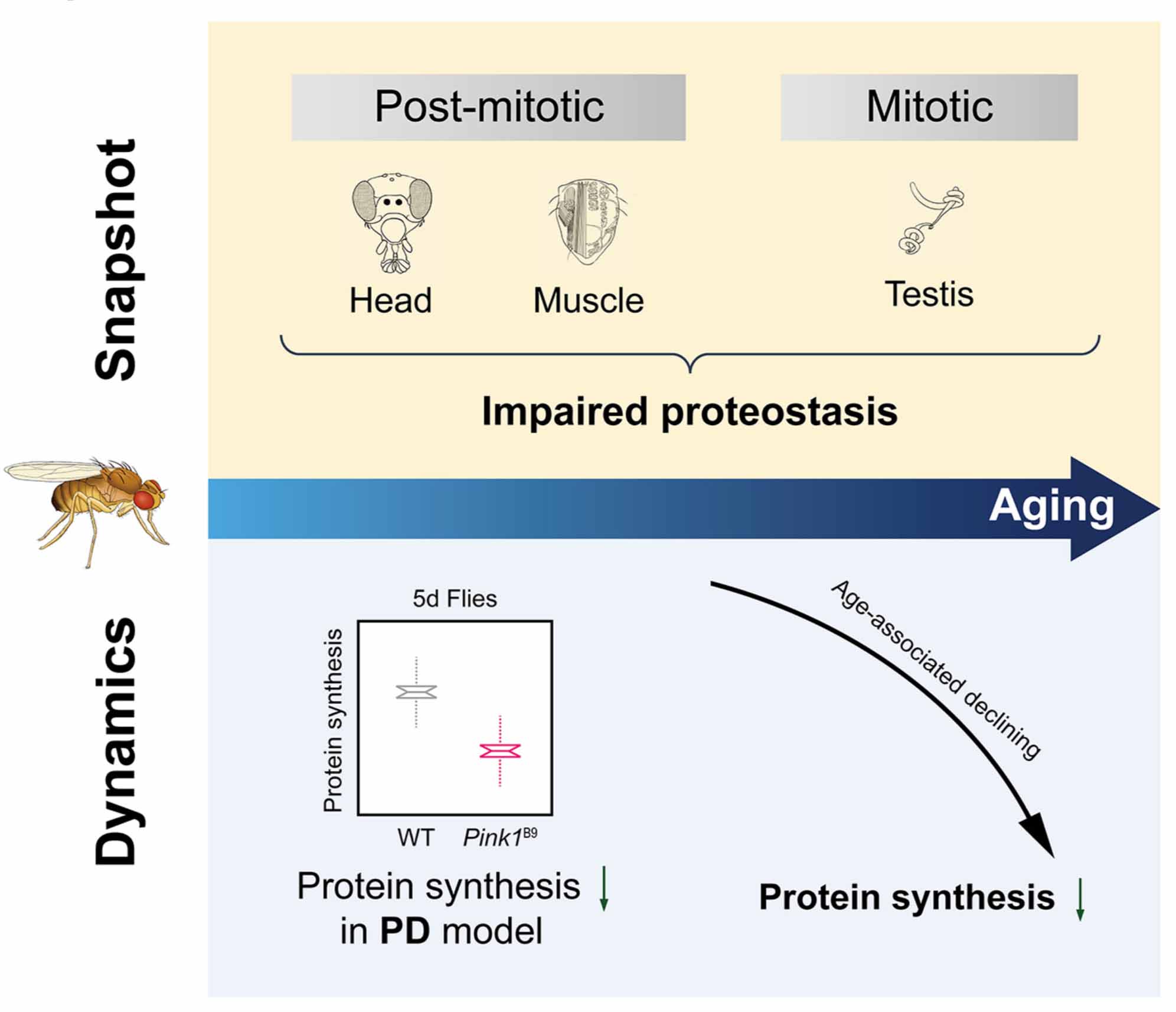
MCP paper Published: Multidimensional Proteomics Identifies Declines in Protein Homeostasis and Mitochondria as Early Signals for Normal Aging and Age-associated Disease in Drosophila
最新的ubH2A文章被F1000推荐
July 26th, 7:30 AM 2019
最新的ubH2A文章被F1000推荐
The first protocol for Drosophila research has now been published
June 21th, 11:30 AM 2019
The first protocol for Drosophila research has now been published. We are proud to contribute.
祝贺马在军博士获得中国科学院大学院长奖学金
June 2nd, 9:00 AM 2019
刘南课题组马在军同学于2014年入学,期间研究工作揭示组蛋白表观修饰在衰老中的变迁和功能。2019年,马在军获得细胞生物学博士学位,同年获得国科大院长奖学金。
Nature Communications Published
May 21th, 9:00 AM 2019
The long-lived proteome constitutes a pool of exceptionally stable proteins with limited turnover. Previous studies on ubiquitin-mediated protein degradation primarily focused on relatively short-lived proteins; how ubiquitylation modifies the long-lived proteome and its regulatory effect on adult lifespan is unclear. Here we profile the age-dependent dynamics of long-lived proteomes in Drosophila by mass spectrometry using stable isotope switching coupled with antibody-enriched ubiquitylome analysis. Our data describe landscapes of long-lived proteins in somatic and reproductive tissues of Drosophila during adult lifespan, and reveal a preferential ubiquitylation of older long-lived proteins. We identify an age-modulated increase of ubiquitylation on long-lived histone 2A protein in Drosophila, which is evolutionarily conserved in mouse, monkey, and human. A reduction of ubiquitylated histone 2A in mutant flies is associated with longevity and healthy lifespan. Together, our data reveal an evolutionarily conserved biomarker of aging that links epigenetic modulation of the long-lived histone protein to lifespan.
frontiers in Genetics Published
May 9th, 9:00 AM 2019
FOXO transcription factors have long been associated with longevity control and tissue homeostasis. Although the transcriptional regulation of FOXO have been previously characterized (especially in long-lived insulin mutants and under stress conditions), how normal aging impacts the transcriptional activity of FOXO is poorly understood. Here, we conducted a chromatin immunoprecipitation sequencing (ChIP-Seq) analysis in both young (2-week-old) and aged (5-week-old) wild-type female fruit flies, Drosophila melanogaster, to evaluate the dynamics of FOXO gene targeting during aging. Intriguingly, the number of FOXO-bound genes dramatically decreases with age (from 2617 to 224). Consistent to the reduction of FOXO binding activity, many genes targeted by FOXO in young flies are transcriptionally altered with age, either up-regulated (FOXOrepressing genes) or down-regulated (FOXO-activating genes) in adult head tissue. In addition, we show that many FOXO-bound genes in wild-type flies are unique from those in insulin receptor substrate chico mutants. Distinct from chico mutants, FOXO targets specific cellular processes (e.g., actin cytoskeleton) and signaling pathways (e.g., Hippo, MAPK) in young wild-type female flies. FOXO targeting on these pathways decreases with age. Interestingly, FOXO targets in aged flies are enriched in cellular processes like chromatin organization and nucleosome assembly. Furthermore, FOXO binding to core histone genes is well maintained at aged flies. Together, our findings provide new insights into dynamic FOXO targeting under normal aging and highlight the diverse and understudied regulatory mechanisms for FOXO transcriptional activity.
Nature Communications Published
Apr 5th, 10:00 AM 2019
Large-scale metabolite annotation is a challenge in liquid chromatogram-mass spectrometry (LC-MS)-based untargeted metabolomics. Here, we develop a metabolic reaction network (MRN)-based recursive algorithm (MetDNA) that expands metabolite annotations without the need for a comprehensive standard spectral library. MetDNA is based on the rationale that seed metabolites and their reaction-paired neighbors tend to share structural similarities resulting in similar MS2 spectra. MetDNA characterizes initial seed metabolites using a small library of MS2 spectra, and utilizes their experimental MS2 spectra as surrogate spectra to annotate their reaction-paired neighbor metabolites, which subsequently serve as the basis for recursive analysis. Using different LC-MS platforms, data acquisition methods, and biological samples, we showcase the utility and versatility of MetDNA and demonstrate that about 2000 metabolites can cumulatively be annotated from one experiment. Our results demonstrate that MetDNA substantially expands metabolite annotation, enabling quantitative assessment of metabolic pathways and facilitating integrative multi-omics analysis.
Journal of Proteome Res. published
Apr 4th, 10:00 AM 2019
The CRISPR-Cas9 system is a genomic editing tool widely used in basic research and under investigation for potential applications in gene therapies for human diseases. To accomplish genomic editing, the system requires the expression of a prokaryotic DNA endonuclease enzyme, Cas9, in host cells. Previous studies have mainly focused on the specificity of Cas9 on the host genome, and thus it is unclear whether this bacterium-derived enzyme affects the protein homeostasis of host cells. Here we applied multi-omic analyses, including transcriptome, proteome, phosphoproteome, Cas9- associated protein interactome, protein synthesis, and histone epigenetic modification, to investigate the cellular response of human cells upon the expression of Cas9. We demonstrate that Cas9 has minimal impact on host cells. Our assessment of intracellular effects of Cas9 paves a path for its broad applications in biological studies and potential clinical translations.
academic report
Jun 28th, 10:00 AM 2018
2018年6月28日,美国University of Pennsylvania, Professor of Biology, Dr. Dejian Ren应刘南研究员邀请,在中国科学院生物与化学交叉研究中心进行了题为“ Ion Channels in Lysosomal Function”的学术报告。Dr.Ren本科学习物理专业,毕业后转入生物领域进行离子通道方面的研究;近年来专注于细胞器离子通道的结构与功能,并持续高产地发表了一系列高水平研究成果,令人印象深刻。报告中,Dr.Ren揭示,细胞器Lysosome表面实际存在多种离子通道,包括Na+, K+, Cl+等离子通道,这些离子通道在感受胞内胞外环境,调控细胞营养供给,清除胞内垃圾等方面起着重要的作用。值得一提的是,一种新型的Lysosomal K+通道蛋白TMEM175,有GWAS证据指出其与神经退行性疾病的发生存在关联,这暗示着Lysosomal离子通道有可能成为治疗这种疾病的药物靶点。
eLIFE PAPER PUBLISHED
May 29th, 10:00 AM 2018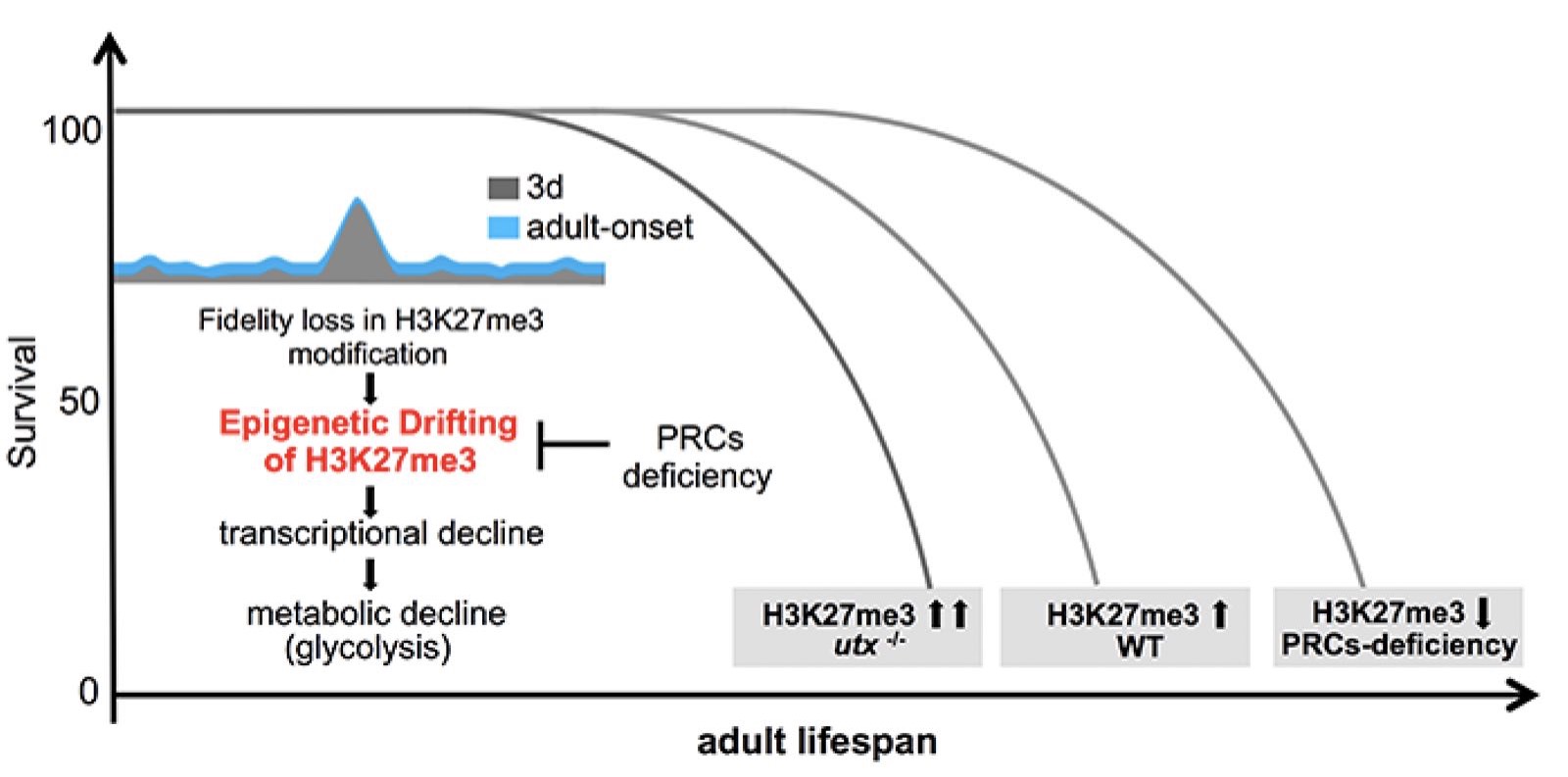
Epigenetic alteration has been implicated in aging. However, the mechanism by which epigenetic change impacts aging remains to be understood. H3K27me3, a highly conserved histone modification signifying transcriptional repression, is marked and maintained by Polycomb Repressive Complexes (PRCs). Here, we explore the mechanism by which age-modulated increase of H3K27me3 impacts adult lifespan. Using Drosophila, we reveal that aging leads to loss of fidelity in epigenetic marking and drift of H3K27me3 and consequential reduction in the expression of glycolytic genes with negative effects on energy production and redox state. We show that a reduction of H3K27me3 by PRCs-deficiency promotes glycolysis and healthy lifespan. While perturbing glycolysis diminishes the pro-lifespan benefits mediated by PRCs-deficiency, transgenic increase of glycolytic genes in wild-type animals extends longevity. Together, we propose that epigenetic drift of H3K27me3 is one of the molecular mechanisms that contribute to aging and that stimulation of glycolysis promotes metabolic health and longevity.
academic report
Jan 10th, 10:00 AM 2018
2018年1月10日,中科院上海上海生科院巴斯德所研究所潘磊研究员应刘南研究员的邀请,在中国科学院生物与化学交叉研究中心进行了题为“The homeostasis of intestinal innate eco-immunology ”的学术报告。潘磊研究员主要介绍了利用果蝇构建了成熟的病原菌感染模型,并利用大规模遗传筛选体系,寻找到Bap180因子具有调节肠道天然免疫平衡的新功能。Bap180应答肠道的IMD天然免疫通路,以负反馈方式在转录水平上限制IMD通路的过度活化,同时抑制由促炎因子Eiger介导的肠道炎性风暴和肠上皮组织损伤。小鼠肠上皮特异敲除进一步证实了Baf180在高等动物中具有同样的保守机制。此外,该研究还表明Bap180不仅针对病原菌急性感染所造成的炎性风暴有抑制作用,同时对于共生菌所触发的肠道慢性炎症的积累也具有重要的平衡作用。该研究中发现了一个重要的监控机体肠道天然免疫平衡的调节因子Bap180/Baf180,并解析了其可以作为关联天然免疫通路和炎性反应的关键节点,为今后肠炎诊断或是药物靶点的开发提供了重要的理论依据。
博士研究生江佳稀答辩完成
Nov 17th, 12:00 AM 2018
刘南课题组江佳稀同学成功的完成了博士答辩。江佳稀同学所完成的学位论文:微小RNA在衰老炎症中的机制研究,具有创新的理论意义。其主要的创新性有:1)发现了新的调控衰老的微小RNA及作用靶mRNA基因;2)微小RNA及靶mRNA基因在果蝇,小鼠及人中高度保守,其调节衰老炎症的作用皆为首次发现;3)人衰老脑组织中,靶基因表达异常上调,与衰老进程相关。答辩委员会以无记名投票的形式一致同意通过该同学的博士学位论文答辩,建议授予江佳稀同学理学博士学位。恭喜江佳稀同学。
academic report
Nov 6th, 16:00 PM 2017
2017年11月6日,美国Baylor College of Medicine,美国HHMI研究员 Dr. Meng Wang(王萌)应陈椰林研究员和刘南研究员的邀请,在中国科学院生物与化学交叉研究中心进行了题为”Metabolic Drivers in Longevity Regulation”的学术报告。王萌研究员课题组以线虫为模型,聚焦机体关键的内分泌器官(脂肪组织和生殖系统)在衰老中的调节作用。并作出一系列新发现,因此获得Ellison Medical Foundation青年学者奖,美国细胞生物学会Gibco新兴领袖奖,美国国立卫生研究院先锋奖,Glenn衰老生理机理奖等众多奖项。在本次报告中,王萌研究员主要介绍了脂质作为信号分子介导细胞内和细胞间的信号传导,进而调控衰老的相关工作。在脂肪细胞内,油酰乙醇胺 (OEA)在分子伴侣LBP-8的帮助下可以介导溶酶体到细胞核的反向信号转导通路,调控衰老。在肠道中,细菌分泌的多糖colanic acid可以通过调控宿主线粒体功能,进而调控衰老。这两项研究揭示了代谢产物在衰老调控中的重要作用。更重要的是,王萌研究员课题组基于stimulated Raman scattering (SRS) microscopy平台,以定量在体观察不同脂质分子动态变化,并以此为基础大规模筛选了调控脂质代谢的基因,为后续研究提供了有力的创新平台.
博士研究生王辉答辩完成
May 22nd, 14:00 PM 2017
刘南课题组王辉同学成功的完成了博士答辩。王辉同学所完成的题为果蝇成体及衰老过程中RNA加工和表观遗传学机制研究的学位论文,具有创新的理论意义。其主要的创新性有:1)发现了Nibbler和Hen1蛋白对piRNA 3’末端的拮抗调节,该长度调节机制影响piRNA对转座子的沉默效率。进一步研究发现piRNA具有Nbr剪切活性依赖的衰老相关调节特征;2)提出了H3K27me3随衰老增加,但增加的修饰表现为准确性下降。因此,组蛋白H3K27me3修饰的准确性缺失可作为新的衰老标志;3)揭示了全基因组范围内H3K27me3信号的扩散性增加,是驱动生物体衰老的全新机制;4)表明了糖酵解代谢通路的激活对维持健康代谢和生物体寿命的重要作用。答辩委员会以无记名投票的形式一致同意通过该同学的博士学位论文答辩,建议授予王辉同学理学博士学位。恭喜王辉同学。
博士研究生马在军获“三好学生”标兵
May 3rd, 16:00 PM 2017
中国科学院大学“三好学生标兵”用以奖励在学习及科研创新中有出色表现,思想品德人格修养方面得到好评的优秀学生。中国科学院生物与化学交叉研究中心从已获得“三好学生”和“优秀学生干部”荣誉称号的学生中推选,授予马在军同学2016-2017年度“三好学生标兵”荣誉称号。该同学自2014年入学以来,一直秉承踏实努力、认真学习的态度,以优异的成绩通过了所修课程,学习掌握了较为扎实的专业知识。凭借优良表现已获得2015-1016学年“三好学生”称号。科研上,该同学相信勤能补拙,在老师同学的帮助下,经过不断地尝试探索,交流合作。从表观组,转录组,蛋白组,代谢组四个层面系统全面的研究了多梳蛋白复合物调控果蝇衰老的机制,相关工作已进入整理写作阶段。以此的研究为基础,将在小鼠中进行验证性研究。下一步将借助计算生物学、单细胞测序等手段揭示衰老机制。
交叉中心博士研究生王辉荣获2016年度国家奖学金
Jan 9th, 14:00 PM 2017
经过中国科学院生物与化学交叉中心全体PI遴选,中心内部公示以及中国科学院大学公示,交叉中心博士研究生王辉同学荣获2016年度国家奖学金。研究生国家奖学金用于奖励表现优异的全日制研究生,自2012年起,每年评选一次。王辉同学是2012级研究生,并于2013年进入刘南老师课题组攻读博士学位。在学习课程上,所有课程均通过且成绩优异。在科研工作上,充分思考,积极探索。目前,分别以第一作者在Development (IF: 6.059) 和第三作者在Cell Reports (IF: 7.87) 发表研究型论文。其中第一作者文章“Antagonistic roles between Nibbler and Hen1 modulate piRNA 3’ ends in Drosophila” 揭示了果蝇中Nbr和Hen1蛋白对piRNA 3’ 端的拮抗调节机制,证实这种长度调节与piRNA的功能具有相关性,并首次报道piRNA在衰老过程中被有序调控。在进一步的研究工作中,王辉同学揭示了表观遗传与衰老的调节机制,整合表观组,转录组,蛋白组及代谢组多维数据,相关工作正在整理,预期投稿到顶尖学术期刊。交叉中心在国家奖学金的评选中,坚持以学术成果为导向,公开,透明。交叉中心鼓励研究生积极攻坚克难,追求卓越,发表更多高水平的,突破性的成果。
academic report
July 22th, 14:00 PM 2016
2016年7月22日中国科学院上海生命科学院研究员李党生博士访问我组及交叉中心,并做学术报告,题目为”How to publish in high profile journals”. 李党生博士现为我国自主创办的学术杂志“Cell Research”和“Cell Discovery”执行总编。在李党生博士的带领下,这两个自主期刊,尤其是“Cell Research”实现跨越式发展,在10年内,将影响因子从3一路提升至15,已成为生命科学领域的高端期刊。在发表高水平文章的要素上,李党生博士提到了:1. conceptual advance (ask a key, fundamental question); 2. solid supportive data; 3. physiological relevance。在成为合格博士的问题上,李党生博士提到了:1. 建设性思维;2. 批判性思维。 李党生博士的报告着实精彩,发人深省。
academic report
June 3rd, 13:30 PM 2016
2016年6月3日,我们迎来了四位专家到访并做学术报告,分别是:王志萍教授(浙江大学,报告题目:Protein quality control and the health of neurons); 齐瀛川教授(杭州师范大学,报告题目:Cleavable ER-bound transcription factors Myrf-1 and Myrf-2 cooperatively controls the synaptic rewiring in C. elegans);丁梅研究员(中科院遗传与发育研究所,报告题目:Synaptic maintenance and function in C. elegans);黄勋研究员(中科院遗传与发育研究所,报告题目:Lipid storage: from organelle to tissues)。报告会的内容涉及到神经生物学和细胞生物学相关领域,从分子水平的蛋白稳态调控,脂类代谢调控,到细胞水平的神经突触形成及可塑性。
academic report
April 26th, 13:30 PM 2016
2016年4月26日下午2点,中国科学院广州生物医药与健康研究院潘光锦研究员到访,并做学术报告。潘光锦研究员首先做了院情介绍,阐述了IPS研究的基础、应用及未来发展的趋势。此外,潘光锦研究员介绍了他课题组团队利用人体尿液分离诱导生成IPS的研究。
academic report
December 18th, 9:30 AM 2015
WeillMedical College of Cornell University Dr. Chao Zhang应刘南研究员的邀请,在中国科学院生物与化学研究中心进行了“Induced pluripotent stem cells for the study of cellular aging and rejuvenation”的学术报告。报告中,张博士在背景部分说明了iPSc对于神经和衰老疾病研究的重要意义,随后介绍了目前诱导性多能干细胞处理过程中的一些挑战,最后张博士引用大量文献阐述了目前通过二代测序技术在转录组、表观遗传修饰等等方面对衰老标志进行的探究。张博士专注于二代测序技术的数据分析研究,试图在结合转录组、表观遗传修饰以及更多的层面上,找到衰老相关标志。张博士的精彩报告让大家对如何通过二代测序及细胞重编程技术探究衰老标志有了深刻的认识。师生踊跃提问,张博士详细解答并与大家深入探讨交流。此次报告圆满结束。
academic report
May 4th, 10:00AM 2015
2015年5月4日,加尼福尼亚大学的舒校坤博士在中国科学院生物与化学交叉研究中心进行了题为“Arationally designed fluorogenic protease reporter visualizes spatiotemporal dynamics of apoptosis in vivo”的精彩学术报告。本次报告由刘南研究员主持。本次报告主要的内容是关于一种新的成像技术。基于荧光共振能量传递的reporters已经广泛用于细胞信号的成像。但是,由于在体内应用中信号较弱,导致了一定的缺陷。荧光reporters解决了这个问题,目前还没有这类的蛋白酶被证明可以在体内成像。现在他们重新设计了一个红外荧光蛋白,蛋白酶的活性可以调节发色团的掺入。基于蛋白酶的活化,红外荧光蛋白酶reporter在没有外源性辅助因子的情况下也可以发光。为了证实这一生物学的应用,他们设计了一个infrared fluorogenic executioner-caspase reporter,它在细胞凋亡和胚胎形态中起着时空调节的的作用,同时也在果蝇肿瘤发生过程中的凋亡动力学中起着相同的作用。这种设计的支架将会应用于其他蛋白酶类的engineer reporters中,但是必须具有特异性切割序列。他们目前的工作展示了一个新的方法,主要是通过处理特异性的蛋白与发色团之间的相互作用而达到。报告结束后,师生们踊跃提出自己的疑问与见解,舒博士为大家一一作答,并进行了进一步的探讨和交流。最后在大家的掌声中报告圆满结束。
academic report
August 11th, 10:00AM 2014
2014年8月11日来自美国哈佛大学George Church实验室的古良才博士在中国科学院生物与化学交叉研究中心王召印研究员的主持下进行了题为“Protein Barcoding & In Situ Sequencing for Multiplex Single-Molecule Proteomics ”的学术报告。古良才博士于1998年兰州大学化学专业取得理学学位后,于2002年清华大学生物化学专业获得理学硕士学位,随后前往美国密歇根大学,于2008年取得药 物化学专业博士学位。现就职于哈佛大学医学院,在G.M.Church教授实验室担任research fellow。古良才博士是十分优秀的多领域复合型人才,获得过众多奖项,发表了数十篇文章,他致力于将新的基因测序技术single molecular interaction-sequencing(SMI-Seq)技术不断完善,并且运用于多领域,尤其是药物筛选中。在报告中,古博士为我们讲述了基因测序的发展,报告内容生动而详实的介绍了新的基因测序技术SMI-Seq的原理以及对未来应用的展望。古博士精彩的报告使交叉中心的同学收获颇丰,师生们积极提问,就感兴趣的问题古博士进行了深入的交流,大家纷纷表示获益匪浅。







